1. What are your differential diagnoses for anaemia?
Anaemia is divided in to two major categories: regenerative (loss of blood) and non-regenerative (lack of blood production). It may take up to 5-7 days for dogs to show a regenerative response after blood loss, this group may be called ‘pre-regenerative’.
Differential diagnoses for regenerative anaemia:
Haemorrhage - cavitary bleeding (e.g. abdomen, thorax), gastrointestinal (always check faecal colour), renal/urinary loss, respiratory tract (e.g.epistaxis), oral cavity (always check the hard palate in cats to assess for a menrath’s ulcer), bleeding disorders (e.g. thrombocytopenia, coagulopathy), which may result in bleeding in multiple sites.
Haemolysis - immune mediated destruction (which may or may not be associated with an underlying condition – previously termed primary or secondary IMHA), haemophagocytic neoplasia, infectious disease (Mycoplasma, Babesia), toxins (snake evenomation, bee stings), hypophosphatemia (e.g. during diabetic ketoacidosis treatment).
Differential diagnoses for non-regenerative anaemia:
Diseases of the bone marrow - immune-mediated disease (precursor targeted immune anaemia (PIMA), pure red cell aplasia), neoplastic infiltration, bone marrow necrosis, iron deficiency (secondary to chronic blood loss), infectious disease (e.g. FeLV, FIV, Ehrlichia).
Diseases which affect the bone marrow - chronic kidney disease, endocrinopathies (e.g. hypothyroidism), chronic inflammatory/infectious or immune-mediated disease.
2. Would you give a blood transfusion to this patient? What product would you select?
There is no specific ‘number’ (i.e. PCV or HCT) where a blood transfusion should be performed. Patients may cope differentially to their anaemia due to several other factors. For example, patients that lose blood quickly do not have time to adapt to this so will become compromised more quickly when compared to dogs whose blood count drops more slowly. Some patients (e.g. sighthounds) may start with a higher blood count so may be relatively more anaemic at a higher haematocrit when compared to other dogs.
This patient was showing signs of ‘transfusion dependency’ such as tachycardia, hyperdynamic pulses, tachypnoea and severe weakness, so the decision was taken to give a transfusion. A transfusion with packed red blood cells was chosen. A whole blood transfusion would be an alternative option, but would be less ideal due to the increased volume required, thereby increasing the risk of volume overload.
3. What tests would you like to perform next?
A complete blood count, particularly to assess the reticulocyte count, which would be elevated in a regenerative anaemia. Also, to assess for abnormalities of other cell lines (e.g. neutrophils or platelets) which may be seen with some bone marrow diseases.
A slide agglutination is useful supportive evidence for immune mediated haemolytic anaemia. If this is negative, then a Coombs test could be considered.
Biochemistry to assess for hyperbilirubinemia, which may be see in red blood cell destruction, low protein levels (which may be low in haemorrhage or high in chronic inflammation), to assess for signs of endocrinopathies or renal disease.
Bicavitary imaging to assess for cavitary bleeding should be performed promptly to assess for evidence of cavitary haemorrhage, which we would want to identify quickly.
4. Can you interpret these slide agglutination tests?
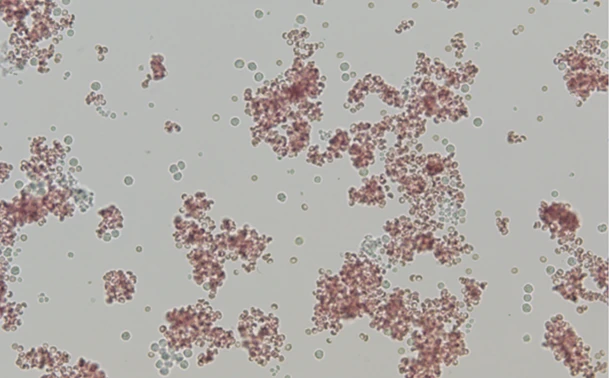
Slide agglutination: one drop of blood in 4 drops of saline
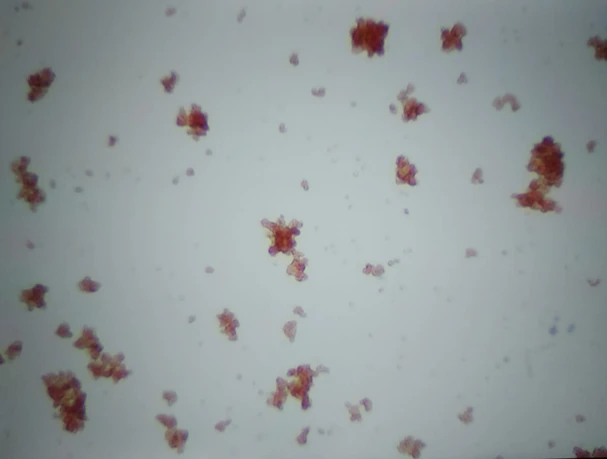
Slide agglutination: one drop of blood in 49 drops of saline
These cells are agglutinating, this is a positive result. The cells appear to be sticking together in a classic ‘bunches of grapes’ appearance. It is recommended to always interpret a slide agglutination test under the microscope, rather than macroscopically. Diluting blood to 49:1 dilution increases the specificity to diagnose IMHA2 , so the author also does this when the 4:1 dilution is positive.
A little later, the results for your patient are returned:
Complete Blood count:
- RBC 2.39 (5.65-8.87 x 10^12/L)
- Haematocrit 0.14 (0.37-0.61 L/L)
- Haemoglobin 53 (131-205g/L)
- MCV 61.6 (61.6-73.5fL)
- MCH 22.2 (21.2-25.9pg)
- MCHC 361 (320-379g/L)
- Reticulocytes 185.2 (10-110K/uL)
- WBC 15.2 (5.05-16.76x10^9/L)
- Neutrophils 11.37 (2.95-1164 x 10^9/L)
- Platelets 159 (148-484x10^9/L)
Smear evaluation: Marked spherocytosis observed.
Biochemistry:
- Globulin 46 (25-45g/L)
- Albumin 29 (22-39g/L)
- Total protein 75 (52-82g/L)
- ALT 32 (10-125 U/L)
- ALP 225 (23-212 U/L)
- Bilirubin 19 (0-15 Umol/L)
- Cholesterol 4 (2.84-8.26 mmol/L)
- Glucose 5.39 (3.69-7.95 mmol/L)
- Creatinine 49 (44-159 umol/L)
- Urea 6.2 (2.5-9.6 mmol/L)
- Phosphorous 1.11 (0.81-2.2 mmol/L)
- Calcium 2.36 (1.98-3.00 mmol/L)
- Sodium 155 (144-160 mmol/L)
- Potassium 3.6 (3.5-5.8 mmol/L)
- Chloride 109 (109-122 mmol/L)
Point of care abdominal and thoracic ultrasound:
No free fluid found in either cavity.
5. Can you reach a diagnosis?
Are results are consistent with a diagnosis of IMHA. When we follow the useful algorithm (see below) from the consensus guidelines on diagnosis of IMHA1, we see we have sufficient signs of immune mediated red cell destruction (spherocytes and a positive slide agglutination test) and signs of haemolysis (hyperbilirubinemia) to reach this diagnosis. A further suspected clue that haemolysis is occurring can be detected on our haematology results – the haemoglobin is relatively higher than the haematocrit suggesting free haemoglobin. A rule of thumb to follow is that the haemoglobin value should be around three times the haematocrit. So, in this case 14 x3 = 42, which is lower than the haemoglobin value of 53.

Taken from ‘Garden et al. ACVIM consensus on the diagnosis of immune-mediated haemolytic anaemia in dogs and cats. JVIM 2019’
Further tests were then performed to look for possible associative diseases. These included point of care tests for infectious diseases (Angiostrongylosis, Ehrlichiosis, Anaplasmosis), thoracic radiographs and urinalysis. No abnormalities were detected, so a diagnosis of non-associative IMHA was presumptively made (previously known as primary IMHA).
The patient was started on immunosuppressive medication (prednisolone and cyclosporine) and thromboprophylaxis (clopidogrel). She responded well to treatment, her immunosuppressive were weaned then stopped over the following 4 months and no relapse has occurred to date.
For further reading:
Will you recommend vaccination of this patient in future?
Are there any drugs that you would avoid in patients that have previously been diagnosed with immune-mediated disease?
References:
1. Garden OA, Kidd L, Mexas AM, Chang YM, Jeffery U, Blois SL, Fogle JE, MacNeill AL, Lubas G, Birkenheuer A, Buoncompagni S, Dandrieux JRS, Di Loria A, Fellman CL, Glanemann B, Goggs R, Granick JL, LeVine DN, Sharp CR, Smith-Carr S, Swann JW, Szladovits B. ACVIM consensus statement on the diagnosis of immune-mediated hemolytic anemia in dogs and cats. J Vet Intern Med. 2019 Mar;33(2):313-334. doi: 10.1111/jvim.15441. Epub 2019 Feb 26. PMID: 30806491; PMCID: PMC6430921.
2. Sun PL, Jeffery U. Effect of dilution of canine blood samples on the specificity of saline agglutination tests for immune-mediated hemolysis. J Vet Intern Med. 2020 Nov;34(6):2374-2383. doi: 10.1111/jvim.15945. Epub 2020 Nov 10. PMID: 33169867; PMCID: PMC7694812.